CRO Oversight in Australian Clinical Trials: Moving Beyond Delegation to Effective Governance
Australia continues to attract a growing share of global clinical research activity, particularly in early-phase drug development. Efficient start-up timelines, experienced investigators, and strong collaboration between academic institutions and clinical research units make Australia an attractive environment for sponsors seeking to accelerate development programs.
At the same time, the operational model for clinical trials has evolved. Sponsors increasingly rely on Contract Research Organisations (CROs) to manage operational activities including site monitoring, pharmacovigilance, regulatory submissions, and data management. For many biotechnology sponsors, CRO partnerships are essential to conducting trials.
However, outsourcing does not relieve sponsors of regulatory responsibility. Under Australian clinical trial regulations, the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) E6(R2/R3) Good Clinical Practice guidelines, and the Australian National Statement on Ethical Conduct in Human Research (NHMRC, current version), sponsors are ultimately accountable for trial conduct, participant safety, and the integrity of trial data .
Many deficiencies identified during inspections arise not from CRO incompetence but from gaps in sponsor oversight. Sponsors may assume CRO processes function as intended, without having the appropriate systems in place to verify this. This paper explores how such gaps arise, provides detailed inspection-style case studies, and outlines strategies for robust oversight in the Australian context.
The Australian Clinical Trial Environment
Clinical trials in Australia are regulated by the Therapeutic Goods Administration (TGA). Sponsors may conduct trials through either the Clinical Trial Notification (CTN) or Clinical Trial Approval (CTA) pathways. Both require review by Human Research Ethics Committees (HRECs) and adherence to internationally recognised GCP standards (ICH E6(R3)) and the Australian National Statement.
Sponsors remain accountable for trial quality and participant protection, even when operational activities are delegated to external providers. Increasingly, trials involve networks of CROs and specialist vendors, which may include central laboratories, imaging providers, electronic data capture and ePRO systems, and decentralised trial technology providers.
While outsourcing provides expertise and scalability, it also requires governance mechanisms to maintain visibility and control over trial quality and compliance.
The Practical Challenge of CRO Oversight
Oversight failures rarely arise from CRO capability gaps. More commonly, they occur when sponsors rely on assumptions rather than structured verification. Common risks include accepting CRO reports without assessing underlying processes, informal oversight practices lacking documentation, undefined responsibilities between sponsor and CRO, and limited review of high-risk trial activities.
Regulatory inspections highlight these gaps. Sponsors are expected to actively demonstrate oversight and risk management throughout the study lifecycle, consistent with ICH E6(R2/R3) and the Australian National Statement.
The following inspection case studies, based on themes identified through the TGA GCP inspection program, illustrate common oversight gaps when sponsor governance structures are insufficient.
Case Study 1 – Electronic Data System Oversight
A small biotech sponsor outsourced a Phase I trial to a CRO that implemented an electronic patient-reported outcome (ePRO) system.
During inspection, regulators requested documentation showing system validation. The CRO had provided a validation summary, which the sponsor had accepted without review. The audit trail and testing under trial conditions had not been independently verified.
Regulatory Concern: Sponsors are accountable for ensuring that electronic systems used for clinical trial data are validated and compliant with GCP.
Action Taken: Sponsor conducted formal system review, documented oversight decisions, and updated procedures for all future trials.
Lesson: Verification of vendor systems is a sponsor responsibility.
Case Study 2 – Monitoring and Site Eligibility
A multi-centre trial relied on CRO monitoring. An internal audit found participants enrolled outside eligibility criteria at one site. Monitoring reports had documented review but did not reference source documents.
Regulatory Concern: Sponsors must ensure monitoring systems are capable of detecting protocol deviations and ensuring data accuracy and participant safety.
Action Taken: Enhanced oversight included periodic review of monitoring outputs and escalation procedures.
Lesson: Oversight must focus on process quality and the operational milestones.
Case Study 3 – Safety Reporting Across Multiple Vendors
A sponsor outsourced pharmacovigilance to a CRO managing multiple regional trials. Inspection revealed safety review discussions documented only via emails and escalation decisions were not formally recorded.
Regulatory Concern: Sponsors must maintain clear documentation of safety oversight to demonstrate compliance with ICH E6(R3) and Australian GCP guidance.
Action Taken: Implemented formal safety governance with scheduled review meetings, structured documentation, and escalation protocols.
Lesson: Safety oversight requires formal governance structures and must not rely on informal communication.
These case studies define where accountability sits and demonstrates the importance of active sponsor oversight. The key takeaways:
Documentation is critical: Activities must be clearly recorded.
Oversight must be active, not passive: Simply reviewing reports is insufficient.
Responsibilities must be clear: Ambiguity between sponsor and CRO responsibilities creates compliance gaps. Ultimately, accountability remains with the sponsor.
Risk-based focus is a priority: Prioritise oversight of high-risk processes such as participant safety and data integrity.
Strengthening CRO Oversight
Beyond meeting regulatory requirements, there is a competitive advantange for sponsors to take more ownership of CRO oversight. Sponsors that demonstrate strong governance often experience:
Smoother regulatory inspections
Higher investor confidence
More reliable trial data
Stronger CRO relationships
To support sponsors in strengthening their CRO oversight, a structured governance framework should be implemented and include:
Vendor Qualification: Assess CRO quality systems, regulatory history, and operational capacity.
Delegation Clarity: Clearly define tasks, retained accountabilities and responsibilities, and escalation pathways.
Operational Transparency: Maintain access to monitoring reports, deviation logs, and key performance indicators.
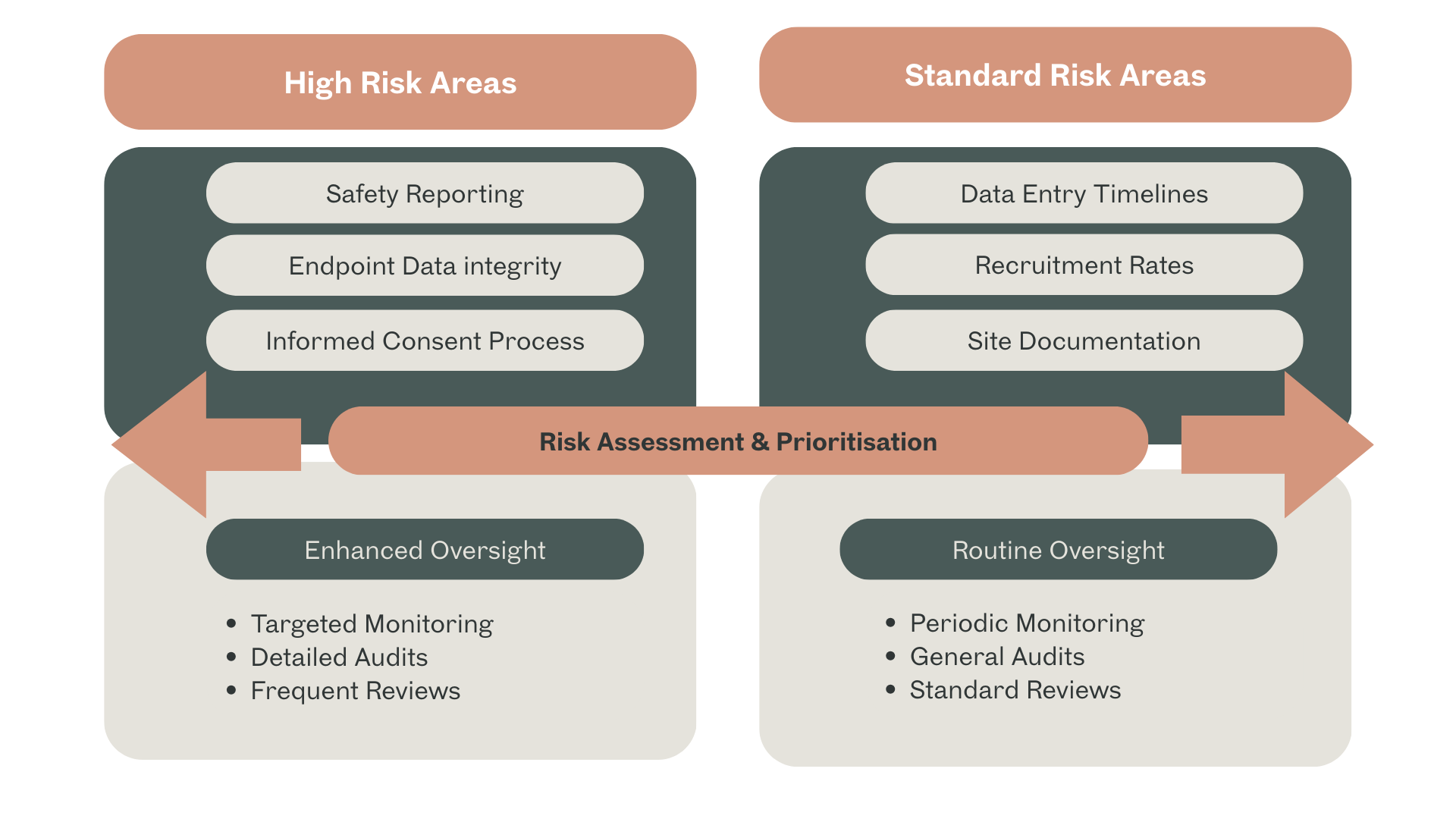
Risk-Based Oversight: Focus on critical data, safety reporting, informed consent, and investigational product accountability.
Figure 1 – Example of Risk-Based Sponsor Oversight Framework: Prioritisation of trial activities by risk: High Risk (Safety Reporting, Endpoint Data, Informed Consent), Medium Risk (Monitoring, Data Entry), Low Risk (Administrative tasks), and associated oversight intensity from Sponsor.
Continuous Governance: Conduct periodic audits, governance meetings, and performance reviews .
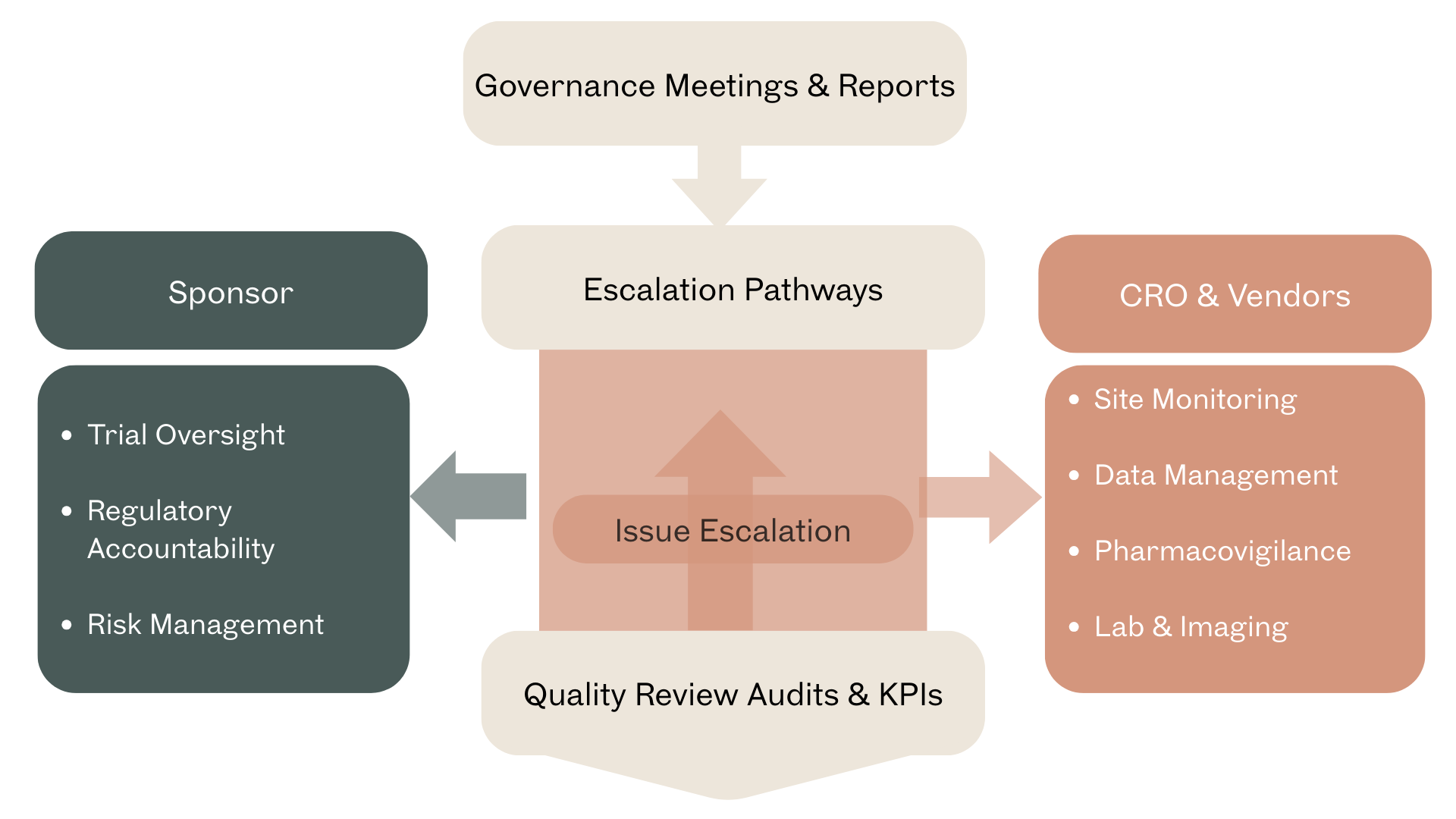
Figure 2 – CRO Oversight Governance Model: A flow diagram showing the sponsor’s oversight responsibilities relative to CRO functions, highlighting review cycles, escalation pathways, and quality control points.
Additional Considerations
CRO oversight is not just about appeasing the regulators and meeting specific Australian requirements. There are financial and reputational implications for inadequate CRO oversight, including:
Research and Development Tax Incentive
A lack of adequate oversight of a CRO can weaken an R&D Tax Incentive (RDTI) claim because the company claiming the incentive must be able to demonstrate that it conducted and controlled the R&D activities, even where operational work was outsourced. The Australian Taxation Office and AusIndustry assess whether the Australian sponsor was accountable for the scientific direction of the research, including the development of the hypothesis, study design, and interpretation of results. If a CRO is effectively designing and running the study with limited sponsor involvement, it may be determined that the sponsor did not conduct the R&D activities, which could result in some or all clinical trial costs being excluded from the RDTI claim.
Maintaining clear evidence of sponsor oversight, such as protocol approval, governance meetings, and review of study data, helps demonstrate that the sponsor retained control of the experimental work and bore the associated financial and technical risk.
Artificial Intelligence
Artificial intelligence (AI) can automate routine oversight tasks such as data reconciliation, query detection, and trend analysis, to reduce manual workload and enable clinical teams to concentrate on critical decision-making and governance activities. While these technologies have the potential to improve efficiency and enable earlier detection of quality issues, regulatory expectations remain clear: AI may support oversight activities, but sponsors retain responsibility for system validation, decision-making, and interpretation of outputs within the framework of Good Clinical Practice.
The Future of Sponsor Oversight
Modern trials increasingly involve digital and decentralised technologies, expanding the vendor ecosystem. Sponsors must oversee not only CROs but also technology providers and specialist service partners. Integrated governance systems and real-time monitoring tools will become essential for managing these complex trial networks while maintaining compliance with ICH E6(R3) and the Australian National Statement.
Conclusion
CROs play an essential role in trial delivery, but outsourcing does not reduce sponsor accountability. Sponsors must actively manage, document, and demonstrate oversight of outsourced activities, with clear governance structures, risk-based monitoring, and effective communication across all vendors. In the Australian regulatory environment, strong CRO oversight safeguards participant safety, ensures data integrity, and supports credible evidence generation.
Want to Know More?
Sunrise Clinical supports sponsors through structured CRO oversight frameworks, governance models, and inspection readiness strategies for clinical trials conducted in Australia.
For further information about our CRO oversight services or regulatory expectations for conducting clinical trials in Australia, please contact:
Justin Jacka
Director and Principal Clinical Research Consultant
Sunrise Clinical
E: justin@sunriseclinical.com.au
M: 0427 968 656
References
• ICH E6(R3) Good Clinical Practice, International Council for Harmonisation, 2022
• Australian National Statement on Ethical Conduct in Human Research (2025), National Health and Medical Research Council
• Therapeutic Goods Administration, Guidance on Preparing for GCP Inspections, 2024
• Therapeutic Goods Administration, Insights from the GCP Inspection Program, 2023–2024